Nucleotides join to form longer molecules
A trinucleotide

Phosphodiester bonds are shown within green boxes.
Two nucleotides can join together by forming another covalent bond between a phosphate group and carbon 3' on the next pentose sugar.
This is then called a
phosphodiester bond, and it is quite a strong linkage but it is flexible and can rotate somewhat so the strand can become curved.
A
polynucleotide is formed when this process is repeated many times.
As a result, the backbone of DNA and RNA is made up from alternating sugar and phosphate groups which are not easily broken apart.
The 3' and 5' labels are important in identifying the ends of nucleic acid strands, and showing the direction of propagation.
This process only works in one direction, adding nucleotides to the 3' end of the developing polynucleotide chain.
In fact the 3' ends of DNA and RNA molecules have hydroxyl groups (-OH), whereas all the other nucleotides have phosphate groups attached at the 3' position.
The diagram at the right could be a short section of RNA, or a single strand of DNA.
Of course DNA has deoxyribose, and thymine not uracil.
Nucleotide nomenclature
Do not confuse the base thymine with thiamine - the vitamin (B1)
The names of, or abbreviations for, the 5 (nitrogenous organic)
bases in nucleic acids are generally quite well known.
Adenine A
Cytosine C
Guanine G
Thymine T
Uracil U
A
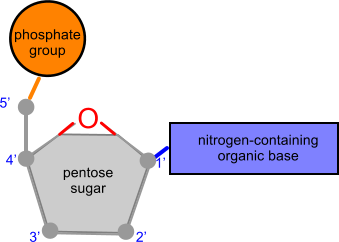
nucleoside normally consists of a nitrogenous base section bonded to a pentose sugar.
These are given slightly different names:
Aden
osine
Cyt
idine
Guan
osine
Thym
idine
Ur
idine
And a
nucleotide consists of a nucleoside attached to a (single) phosphate group.
Adenosine monophosphate
Cytidine monophosphate
Guanosine monophosphate
Thymidine monophosphate
Uridine monophosphate
These could be written as
AMP, CMP, GMP, UMP for the RNA versions - ribonucleotides
( dAMP, dCMP, dGMP, dTMP, for the DNA versions - deoxyribonucleotides)
But some have more than one phosphate group:
ATP , ADP - adenosine triphosphate and diphosphate (and also GTP and GDP) are well known for their role in energy transfer in respiration and metabolic reactions in general, and UTP in glycogen synthesis






 These form antiparallel strands
These form antiparallel strands
 These two strands become gently twisted into a double helix.
These two strands become gently twisted into a double helix.

