Recycling
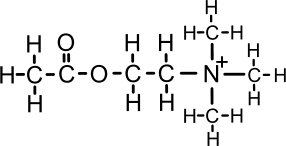
Acetylcholine is broken down by the enzyme
(acetyl)choline esterase. This stops depolarisation.


The breakdown of acetylcholine
Acetic acid (ethanoic acid) can ionise to give acetate (ethanoate) CH3COO-
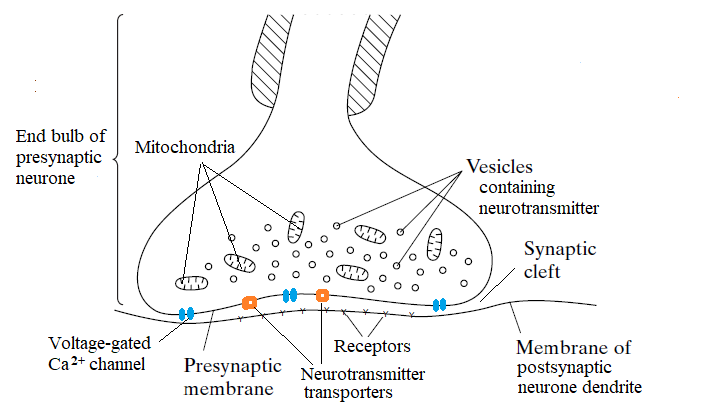
Choline diffuses into the synaptic cleft and is reabsorbed into the presynaptic knob. Here it is (re-)combined with an acetyl group and incorporated into vesicles, ready for use in a subsequent impulse.
Other neurotransmitters are not neutralised by hydrolysis, but they are quickly removed from the synapse and re-packaged into vesicles for re-use
There are specific protein transporter channels in the presynaptic membrane for the re-uptake of neurotransmitter components.
The production of vesicles, and the uptake of more acetylcholine, is a multi-stage energy-requiring process.
Calcium is actively pumped out of the presynaptic end bulb - similarly to the Na
+/K
+ pump which works along the axon.
Sodium is actively pumped out of the postsynaptic cell, restoring its resting potential.
There are a number of mitochondria in the end bulb and these obviously provide ATP for these energy-requiring processes.
Mitochondria can also provide acetyl Coenzyme A which donates the acetyl group for the (re-)formation of acetylcholine.
More about acetylcholine esterase
The enzyme acetylcholine esterase has a well-defined active site, often described as a gorge because of its distinctive shape. It has 2 main regions : the binding site - the 'anionic subsite', which holds the charged choline end of the substrate molecule - and beneath this is a catalytic active site - the 'esteratic subsite', which breaks the ester bond. The hydrolysis of acetylcholine is a very rapid process.
Acetylcholine esterase inhibitors
Other charged molecules can be attracted to the anionic subsite and not be affected by the esteratic subsite. This effectively blocks access to the active site and prevents the breakdown of acetylcholine.
As a result, postsynaptic membranes remain depolarised, nervous impulses may last longer or be stronger, and stimulated muscles remain contracted.
There are a number of symptoms resulting from the effects of acetylcholine esterase inhibitors on muscles of the body, summarised by the mnemonic
SLUDGE.
Insecticides
Organophosphate compounds were found to be very toxic to insects as a result of their (irreversible) inhibition of acetylcholine esterase, the phosphate group occupying the anionic subsite where choline normally binds. There was thought to be a major difference in the toxicity of these compounds to mammals including Man and insects - in fact arthropods in general. Evidently this is a result of minor differences in the metabolism of these animal groups.
A number of these compounds have been marketed as insecticides, also used against
arachnids.
Malathion is the main one used for control of pest insects such as mosquitoes. Others have been replaced on the grounds of danger to human health.
For example: dichlorvos - packaged as Vapona strip, which allowed evaporation of the volatile active ingredient - was considered very effective as a control method for insect pests in (limited) spaces such as shops and houses.
However it was noted that people exposed continuously to it had lower levels of acetylcholine esterase in their blood plasma and often experienced a number of neurological symptoms. It was introduced as an insecticide in 1961 but banned in Europe in 1998. It is still available in some parts of the world.
Nerve agents (nerve gases)
As a consequence of research into organophosphate insecticides by German chemists in the 1930s, a number of compounds (Sarin and Tabun) were found to have effects on Man and these were seen as candidates for use in warfare, but they were not used in World War II. Other more toxic compounds (including 'VX' and various Novichok agents) have been discovered since then by researchers in the USA, UK and Russia. These compounds have regrettably been used in a number of scenarios since.
Medicinal applications

Several acetylcholine esterase inhibitors are used clinically.
Donepezil is a medication used in the palliative treatment of Alzheimer's disease. It is not a cure for Alzheimer�s disease, but it may help slow down the speed of progression of symptoms. There are also a number of possible systemic side effects.
Because there is often a lower concentration of acetylcholine in the brain of a person suffering from this condition, communication between neurones can be less efficient, resulting in memory loss and confusion.
As an acetylcholine esterase inhibitor, donepezil reduces the (natural) breakdown of acetylcholine, which means more is available to attach to receptors, so that more ACh is present and remains attached for longer. This results in a greater influx of sodium ions, so that depolarisation of the post-synaptic membrane is achieved, and an action potential is generated in the receiving neurone, leading to an impulse being propagated.
Neurotransmitter reuptake inhibitors
Other neurotransmitter substances may not be hydrolysed after an impulse has passed but simply removed from the synapse and passed back via the cell membrane into the presynaptic neurone from which they originated. This involves
reuptake channels which may be inhibited by other compounds, leading to similar consequences to acetylcholine esterase inhibitors. This inhibition may be competitive, a result of molecular similarity with the neurotransmitter substance, or an allosteric effect, so that the reuptake inhibitor changes the shape of the recognition site for the neurotransmitter. In each case the neurotransmitter remains in place in the synapse for longer, increasing in concentration and has a greater effect.
For example, serotonin reuptake inhibitors (SRIs) are a class of drugs which block the action of the serotonin transporter (SERT), leading to increased extracellular concentrations of serotonin and an increase in 'serotonergic neurotransmission' in these cells. They may have an antidepressant effect.


 Several acetylcholine esterase inhibitors are used clinically.
Several acetylcholine esterase inhibitors are used clinically.





